As an inhibitor of an inflammatory protein associated with the development of psoriasis, adalimumab shows promise as a therapy for pediatric patients with severe plaque psoriasis. Adalimumab treatment for 16 weeks in children and adolescents with severe plaque psoriasis provides significant improvements compared to methotrexate.
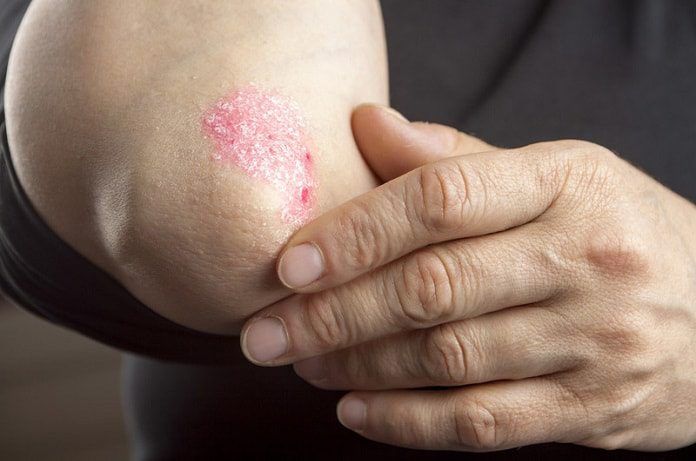
Psoriasis is a chronic skin condition characterized by scales and red patches that are typically found on the scalp, elbows, and knees. This buildup of extra skin cells on the surface of the epidermis is an autoimmune inflammatory disease, which is currently incurable. Immune system T-cells and an abundance of inflammatory protein tumor necrosis factor-alpha (TNF-α) play major roles in the development of psoriasis. Though there are many types of psoriasis, plaque psoriasis is the most common condition which involves the build-up of plaque on the surface of the skin. Itching, burning, soreness, or cracked skin are some of the symptoms associated with the disease and its severity can be classified as mild, affecting less than 5% of the skin’s surface area, moderate, affecting 5 to 10% of the skin, or severe, with more than 10% of the skin’s surface affected. Affecting 2% of the general population, a third of psoriasis diagnoses made by physicians include those who are 20 years of age and younger.
Management of pediatric psoriasis can decrease the risk of psychosocial issues and comorbidities such as, hypertension and diabetes. Initial treatment for patients with limited disease includes topical therapies, while severe pediatric psoriasis is treated using ultraviolet B phototherapy, or systemic treatments, such as methotrexate, ciclosporin, or retinoids. However, though TNF-α inhibitor, methotrexate, has been prescribed to treat children and adolescents, it has not been approved by the European Medicine Agency, thus making it a good candidate for clinical research assessment. Due to the lack of standardized guidelines and approved systemic therapies, managing psoriasis by the blockage of TNF-α, has been challenging. However, in 2015, TNF-α inhibitor, adalimumab, was approved in the United States to treat severe cases in patients who were 4 years of age and older, and who did not respond adequately to topical therapy or phototherapies. Therefore, it is important to compare both inhibitors for their safety and efficacy in treating severe pediatric plaque psoriasis.
A double-blind randomized controlled study was performed to compare the safety and efficacy of adalimumab and methotrexate in children with severe psoriasis. Treatment groups consisted of a total of 114 patients who were randomly assigned to receive either 0.8 mg/kg of adalimumab, 0.4 mg/kg of adalimumab, or 0.1-0.4 mg/kg of methotrexate. Adalimumab was given subcutaneously every other week, whereas, methotrexate was taken orally once weekly. The study consisted of four periods; identified as the 16-week primary treatment, up to 36-week withdrawal, 16-week re-treatment, and 52-week long-term follow-up. Measurements based on the Psoriasis Area and Severity Index (PASI) assessed the percentage of skin affected and 75% improvement, PASI75, was a study endpoint. The Physician Global Assessment (PGA), which measures psoriasis activity, was used to identify clear or minimal areas. At week 16, PASI75 was achieved in 58% of the patients receiving 0.8mg/kg of adalimumab, in 44% of patients receiving 0.4 mg/kg of adalimumab, and in 32% of patients taking methotrexate. Results from the PGA showed 61% of patients receiving 0.8mg/kg of adalimumab, 41% of patients receiving 0.4 mg/kg adalimumab, and 41% of patients taking methotrexate had a clear or minimal PGA score. Initial treatments resulted in adverse events, such as infections for 45% of the patients receiving 0.8 mg/kg of adalimumab, in 56% of the patients receiving 0.4 mg/kg of adalimumab, and in 51% of those taking methotrexate. Compared to methotrexate, treatment with adalimumab in children and adolescents with severe plaque psoriasis provided significant improvements in PASI75. Although there was an increase in the number of patients with a clear or minimal PGA score in the adalimumab group compared to methotrexate, these results did not reach statistical significance. Overall, adalimumab was found to be more effective than methotrexate, with a rapid response and similar safety profile after 16 weeks.
This study is one of few investigations that characterize the long-term safety of treatment of severe psoriasis in children. Though a limitation of the study was a lack of methotrexate control data to compare to the investigated population, the safety and efficacy profile of adalimumab was successfully evaluated for comparison to methotrexate. In conclusion, treatment with 0.8 mg/kg of adalimumab for 16 weeks in children and adolescents with severe plaque psoriasis provided significant improvements in PASI75 and a non-significant increase in patients who achieved clear or minimal PGA compared with methotrexate. These findings provide new insight and an additional option for safe and effective therapy of severe plaque psoriasis in a young population.
Written By: Viola Lanier, Ph. D., M. Sc.